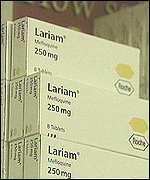
| By Admin1 (admin) on Thursday, June 06, 2002 - 10:22 am: Edit Post |
The Lariam Files


| By Nancy E. Tongue on Sunday, January 26, 2003 - 11:18 pm: Edit Post |
I have had a Peace Corps illness unrelated to Larium but related to my "service" and have fought tooth and nail for medical coverage for 18 years with the system constantly denying me although I have documentation to prove my illness. I am desperately looking for other RPCV's who have gotten sick and mistreated subsequently by the Peace Corps so that we can "go public" or makes some changes. Please email me in NYC at: Fourdirect@aol.com (Nancy)